26 Regulatory Submission
The culmination of years of research, billions of dollars in investment, and thousands of patients’ participation comes down to a single question: will regulators approve the drug? The regulatory submission is where sponsors present their case—the accumulated evidence from preclinical studies and clinical trials that, they hope, demonstrates the product is safe and effective.
Different types of applications serve different products and circumstances:
- New Drug Application (NDA): the primary vehicle for small-molecule drugs in the United States, containing comprehensive data on chemistry, manufacturing, preclinical studies, and clinical trials
- Biologics License Application (BLA): for biological products such as proteins, vaccines, and cellular therapies, following a similar structure but with additional manufacturing details specific to biologics
- Marketing Authorization Application (MAA): the equivalent submission in the European Union, submitted centrally or through national procedures
- Abbreviated New Drug Application (ANDA): facilitates approval of generic drugs by relying on reference products
- Biosimilar applications: demonstrate similarity to an approved reference biologic
- Supplements and variations: allow sponsors to modify existing approvals for new indications or manufacturing changes
International harmonization has established a standardized format for regulatory submissions known as the Common Technical Document (CTD). The CTD organizes information into five distinct modules, as summarized in Table 26.1.
| Module | Name | Content Description |
|---|---|---|
| 1 | Regional Administrative | Forms, labeling, and jurisdiction-specific materials |
| 2 | Summaries & Overviews | Integrated quality, nonclinical, and clinical summaries |
| 3 | Quality (CMC) | Chemistry, Manufacturing, and Controls documentation |
| 4 | Nonclinical | Reports from toxicology and pharmacology studies |
| 5 | Clinical | Clinical study reports and related data materials |
This standardized modularity allows sponsors to prepare a single “core dossier” (comprising Modules 2 through 5) for submission to multiple global regulatory agencies simultaneously. Only Module 1 requires region-specific adaptation, significantly streamlining the process of bringing a new therapy to multiple international markets.
26.1 Building the Submission
Assembling a major regulatory application is a massive undertaking that requires seamless coordination across multiple specialized functions. Regulatory affairs teams lead the effort, managing the submission strategy and maintaining direct communication with agencies to ensure every legal and technical requirement is met. The clinical development group manages the compilation and interpretation of all clinical data, while CMC specialists document the manufacturing and quality controls that guarantee product stability. Simultaneously, nonclinical researchers provide the foundational toxicology data, and medical writers synthesize these disparate streams into clear summaries that help agency reviewers navigate the massive volume of technical information. The final submission is packaged as an electronic CTD (eCTD), facilitating the secure transmission of thousands of documents to regulatory authorities worldwide.
26.2 Pre-Submission Interactions
Smart sponsors do not simply submit applications and hope for the best. They engage with regulatory agencies throughout development and especially before major submissions.
Pre-IND meetings occur before first-in-human studies to clarify the data needed to support clinical development. End-of-Phase 2 meetings represent a pivotal milestone for discussing Phase III plans, where sponsors present their design and evidentiary standards for agency feedback. Finally, Pre-NDA/BLA meetings (or Pre-MAA in Europe) are conducted before the major submission to confirm that the dossier is complete and free of critical unresolved issues.
These meetings, typically conducted as formal written exchanges or in-person discussions, help align sponsor and agency expectations. Different meeting types have different timelines and objectives, as summarized in Table 26.3. Discovering a major issue before submission is far better than discovering it during review.
26.3 Expedited Regulatory Pathways
In response to the urgent need for treatments for serious conditions, regulatory agencies have established specific pathways designed to accelerate the development and review of promising therapies. These pathways do not lower the evidentiary standard for safety or efficacy; rather, they serve to intensify the interaction between the sponsor and the regulator, and in some cases, allow for the use of surrogate endpoints reasonably likely to predict clinical benefit.
Fast Track designation is intended for drugs that treat serious conditions and fill an unmet medical need. This status allows for more frequent meetings with the FDA and the potential for rolling review, where the sponsor submits completed sections of the application before the entire dossier is finalized. Breakthrough Therapy is a more intensive designation for drugs where preliminary clinical evidence indicates the drug may demonstrate substantial improvement over available therapy on a clinically significant endpoint. Accelerated Approval allows approval based on a surrogate endpoint or an intermediate clinical endpoint that is reasonably likely to predict clinical benefit. Finally, Priority Review represents a commitment by the FDA to aim for a review decision within six months for drugs that would provide significant improvements in the safety or effectiveness of the treatment.
Table 26.2 contrasts these distinct mechanisms. Note that a single drug program may utilize multiple pathways simultaneously (e.g., a Breakthrough Therapy that receives Accelerated Approval and Priority Review).
| Designation | Criteria | Key Benefit |
|---|---|---|
| Fast Track | Serious condition + unmet need | Rolling review, frequent communication |
| Breakthrough | Serious condition + substantial improvement | Intensive guidance, organizational commitment |
| Accelerated Approval | Serious condition + surrogate endpoint | Approval based on surrogate (requires confirmation) |
| Priority Review | Significant improvement | 6-month review clock (vs 10 months) |
| Meeting Type | Purpose | Examples | FDA Response Goal |
|---|---|---|---|
| Type A | Critical path issues; Stalled development | Dispute resolution; Clinical hold discussion | 30 days |
| Type B | Key development milestones | Pre-IND, End-of-Phase 2, Pre-NDA/BLA | 60 days |
| Type C | General guidance | Protocol design review; New indication discussion | 75 days |
26.4 The Review Process
Once submitted, applications enter formal review shown in Figure 26.1.
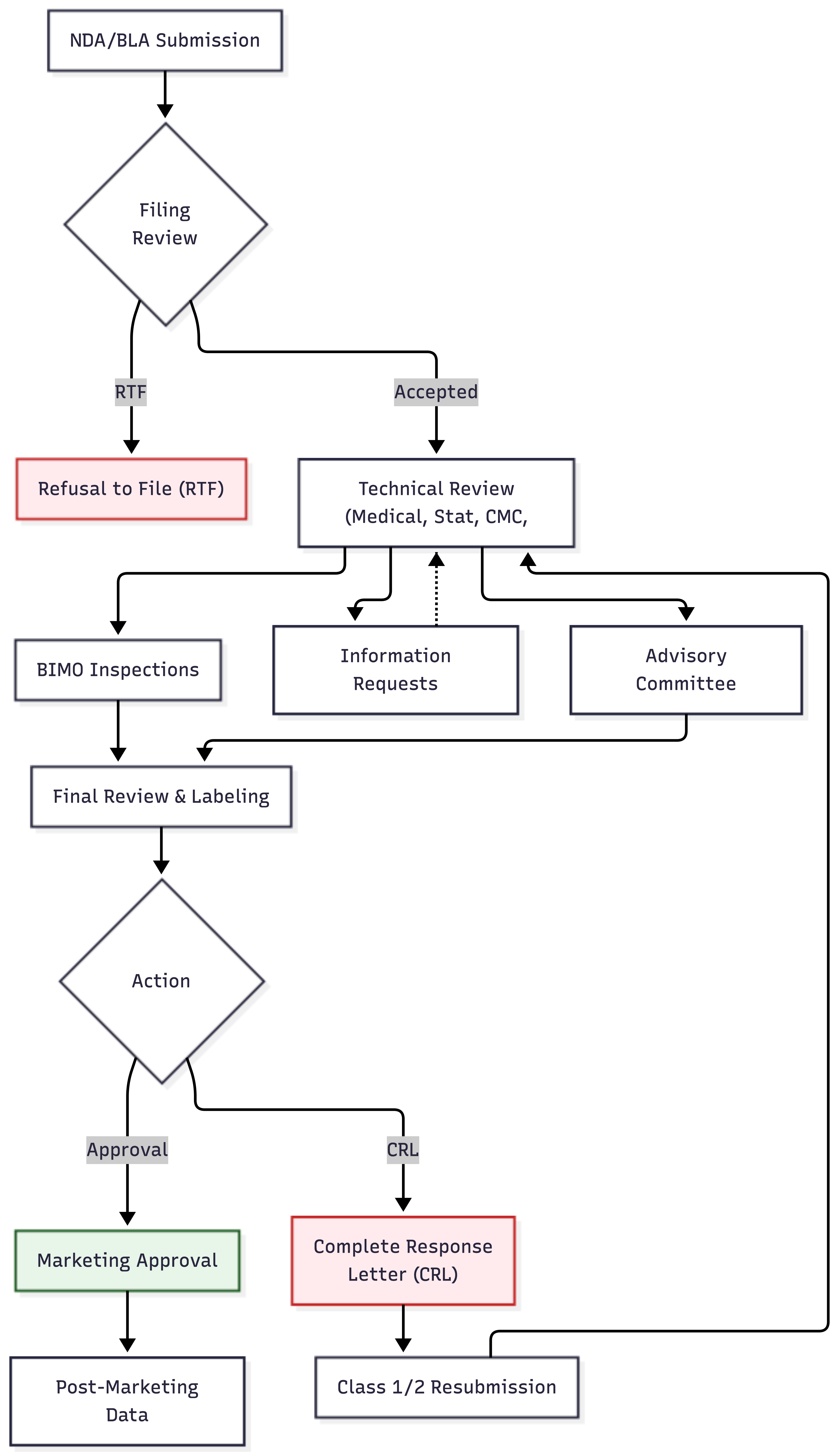
In the United States, FDA first determines whether the submission is complete—whether it contains enough information to permit substantive review. Applications deemed incomplete may be refused to file and returned without review.
If accepted, the application enters a review queue. Review timelines are governed by user fee goals—performance targets established in legislation. Standard review typically takes 10-12 months from acceptance; priority review (for drugs addressing serious conditions with unmet needs) may be completed in 6 months.
Review is multidisciplinary. Medical officers evaluate clinical efficacy and safety. Pharmacologists assess pharmacokinetics. Chemists review manufacturing. Statisticians verify the analyses. Toxicologists examine nonclinical safety.
26.5 Bioresearch Monitoring (BIMO) Inspections
Parallel to the scientific review of the data, the FDA conducts on-site inspections through its Bioresearch Monitoring (BIMO) program to verify the integrity of the data and compliance with regulations. These inspections are a critical stress test of the submission; positive data are worthless if the agency cannot trust that they were collected accurately and ethically.
BIMO inspections typically target clinical sites, the sponsor, and the involved Institutional Review Boards. At clinical sites, inspectors compare source documents against the data submitted in the case report forms, looking for discrepancies or evidence of fraud. Sponsor inspections evaluate the oversight of the trial, checking monitoring reports, data management systems, and safety reporting processes. IRB inspections ensure that ethical oversight was maintained appropriately throughout the study.
Significant findings during a BIMO inspection can result in a Form 483 (listing observations) or a Warning Letter. In severe cases, the FDA may invoke the “Application Integrity Policy,” pausing the review until the data’s reliability can be re-established. Consequently, “inspection readiness” is a continuous activity for clinical operations teams, not just a pre-submission drill.
Reviewers may issue information requests—questions requiring the sponsor to clarify or supplement the submission. Sponsors must respond promptly to avoid delays.
For novel or complex applications, FDA may convene an Advisory Committee—a panel of outside experts who discuss the application in a public meeting and vote on approval recommendations. While advisory committee recommendations are not binding, FDA usually follows them.
26.6 The Decision
At the conclusion of review, FDA issues an action letter.
An approval letter authorizes marketing. The product may now be sold, but usually with specific labeling, potentially with post-marketing requirements or commitments, and subject to ongoing safety monitoring.
A complete response letter (CRL) indicates that the application cannot be approved in its current form. The letter specifies deficiencies that must be addressed. Sponsors may then submit a response, addressing each deficiency. Multiple review cycles may be needed before approval is achieved—or the sponsor may ultimately abandon the product.
Similar decision frameworks exist in other jurisdictions: Europe uses marketing authorization and requests for supplementary information; Japan and other markets have analogous processes.
Regulatory obligations continue well beyond the initial approval. Sponsors are often required to conduct post-marketing requirements (PMRs) or agree to post-marketing commitments (PMCs), which can involve additional safety studies or trials in specialized populations. For therapies with significant identified risks, a Risk Evaluation and Mitigation Strategy (REMS) may be mandated to ensure safe use through communication plans or restricted distribution. Throughout the product’s marketed life, sponsors must maintain vigilant safety monitoring and submit periodic reports to regulatory authorities, ensuring that the benefit-risk balance remains favorable as real-world experience accumulates.